NCERT Exemplar Class 12 Chemistry Chapter 12 Aldehydes Ketones And Carboxylic Acids
NCERT Exemplar for Class 12 Chemistry Chapter 12
NCERT Exemplar for Class 12 Chemistry is an effective study material to boost your knowledge on the various sections of a chapter. Students can rely on this provided exemplar solutions of Chapter 12 Aldehydes Ketones and Carboxylic Acids to solve questions and become more familiar with it. Our subject matter experts at SimplyAcad have made sure to convey all the information and minute details precisely to students for effective learning.
The exemplar contains a total of 46 questions including different types of question patterns such as MCQs based, short answer type, matching type and assertion and reason type answer questions which ensure that students cover every bit of the section. This will raise confidence in them and prepare them for the upcoming 12th board examination. Along with this, there are several NCERT exemplar for class 12 science of all the chapters provided on this platform.
Access Solutions of the NCERT Exemplar Class 12 Chemistry Chapter 12 Aldehydes Ketones And Carboxylic Acids
I. Multiple Choice Questions (Type-I) for Ncert exemplar for class 12 chemistry
1. Addition of water to alkynes occurs in acidic medium and the presence of Hg2+ ions as a catalyst. Which of the following products will be formed on addition of water to but-1-one under these conditions.

Solution:
Option (ii) is the answer.
2. Which of the following compounds is most reactive towards nucleophilic addition reactions?
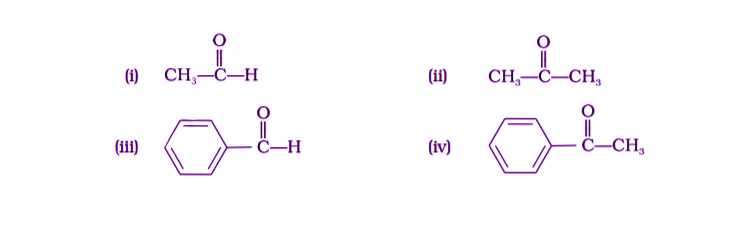
Solution:
Option (i) is the answer.
3. The correct order of increasing acidic strength is _____________.
(i) Phenol < Ethanol < Chloroacetic acid < Acetic acid
(ii) Ethanol < Phenol < Chloroacetic acid < Acetic acid
(iii) Ethanol < Phenol < Acetic acid < Chloroacetic acid
(iv) Chloroacetic acid < Acetic acid < Phenol < Ethanol
Solution:
Option (iii) is the answer.
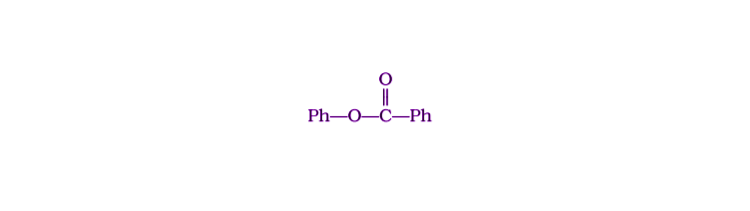
4. Compound can be prepared by the reaction of _____________.

(i) Phenol and benzoic acid in the presence of NaOH
(ii) Phenol and benzoyl chloride in the presence of pyridine
(iii) Phenol and benzoyl chloride in the presence of ZnCl2
(iv) Phenol and benzaldehyde in the presence of palladium
Solution:
Option (ii) is the answer.
5. The reagent which does not react with both, acetone and benzaldehyde.
(i) Sodium hydrogen sulphite
(ii) Phenyl hydrazine
(iii) Fehling’s solution
(iv) Grignard reagent
Solution:
Option (iii) is the answer.
6. Cannizaro’s reaction is not given by _____________.

Solution:
Option (iv) is the answer.
7. Which product is formed when the compound is treated with concentrated aqueous KOH solution?

Solution:
Option (ii) is the answer.
8.

Structure of ‘A’ and type of isomerism in the above reaction are respectively.
(i) Prop–1–en–2–ol, metamerism
(ii) Prop-1-en-1-ol, tautomerism
(iii) Prop-2-en-2-ol, geometrical isomerism
(iv) Prop-1-en-2-ol, tautomerism
Solution:
Option (iv) is the answer.
9.

(i) identical
(ii) positional isomers
(iii) functional isomers
(iv) optical isomers
Solution:
Option (ii) is the answer.
10. Which is the most suitable reagent for the following conversion?

(i) Tollen’s reagent
(ii) Benzoyl peroxide
(iii) I2 and NaOH solution
(iv) Sn and NaOH solution
Solution:
Option (iii) is the answer.
11. Which of the following compounds will give butanone on oxidation with alkaline KMnO4 solution?
(i) Butan-1-ol
(ii) Butan-2-ol
(iii) Both of these
(iv) None of these
Solution:
Option (ii) is the answer.
12. In Clemmensen Reduction carbonyl compound is treated with _____________.
(i) Zinc amalgam + HCl
(ii) Sodium amalgam + HCl
(iii) Zinc amalgam + nitric acid
(iv) Sodium amalgam + HNO3
Solution:
Option (i) is the answer.
II. Multiple Choice Questions (Type-II) for Ncert exemplar for class 12 chemistry
Note: In the following questions two or more options may be correct.
13. Which of the following compounds do not undergo aldol condensation?

Solution:
Option (ii) and (iv) are the answers.
14. Treatment of compound with NaOH solution yields
(i) Phenol
(ii) Sodium phenoxide
(iii) Sodium benzoate
(iv) Benzophenone
Solution:
Option (ii) and (iii) are the answers.
15. Which of the following conversions can be carried out by Clemmensen Reduction?
(i) Benzaldehyde into benzyl alcohol
(ii) Cyclohexanone into cyclohexane
(iii) Benzoyl chloride into benzaldehyde
(iv) Benzophenone into diphenylmethane
Solution:
Option (ii) and (iv) are the answers.
16. Through which of the number of the following reactions of carbon atoms can be increased in the chain?
(i) Grignard reaction
(ii) Cannizaro’s reaction
(iii) Aldol condensation
(iv) HVZ reaction
Solution:
Option (i) and (iii) are the answers.
17. Benzophenone can be obtained by ____________.
(i) Benzoyl chloride + Benzene + AlCl3
(ii) Benzoyl chloride + Diphenyl cadmium
(iii) Benzoyl chloride + Phenyl magnesium chloride
(iv) Benzene + Carbon monoxide + ZnCl2
Solution:
Option (i) and (ii) are the answers
18. Which of the following is the correct representation for intermediate of nucleophilic addition reaction to the given carbonyl compound (A) :

Solution:
Option (A) and (B) are the answers.
III. Short Answer Type
19. Why is there a large difference in the boiling points of butanal and butane-1-ol?
Solution:
Butanal has no intermolecular hydrogen bonding but butan-1-ol has intermolecular hydrogen bonding. This bonding in butan-1-ol makes it more stable at a higher temperature than butanal.
20. Write a test to differentiate between pentan-2-one and pentan-3-one.
Solution:
One can differentiate between pentan-2-one and pentan-3-one by iodoform test. Pentan-2-one have a –CO-CH3 group and therefore forms a yellow precipitate of Iodoform. Pentan-2-one gives a positive iodoform test, whereas, and pentan-3-one does not give a positive iodoform test because they don’t have a –CO-CH3 group.
21. Give the IUPAC names of the following compounds

Solution:
(i) 3-Phenylprop-2-ene-1-al.
(ii) Cyclohexanecarbaldehyde
(iii) 3-Oxopentan-1-al
(iv) IUPAC name: But-2-enal
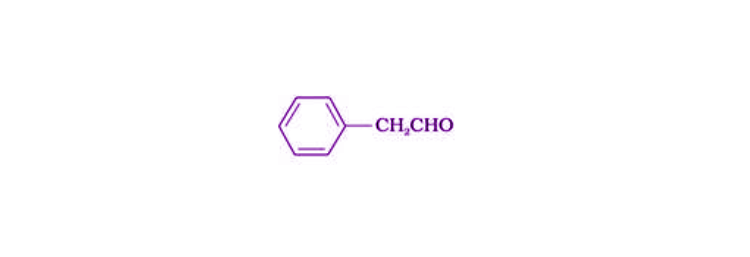
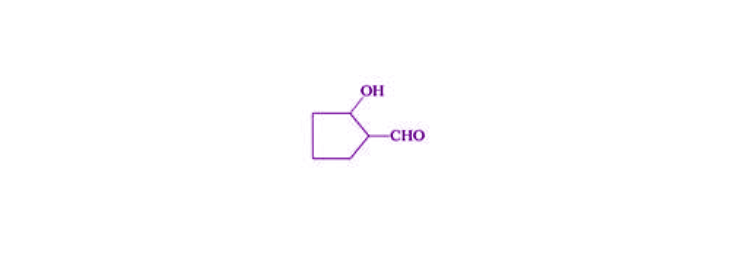
22. Give the structure of the following compounds.
(i) 4-Nitropropiophenone
(ii) 2-Hydroxycyclopentanecarbaldehyde
(iii) Phenyl acetaldehyde
Solution:
(i)

(ii)
23. Write IUPAC names of the following structures.

Solution:
(i) Ethane-1,2-dial.
(ii) Benzene-1, 4-dicarbaldehyde.
(iii) 3-Bromobenzaldehyde.
24. Benzaldehyde can be obtained from benzal chloride. Write reactions for obtaining benzyl chloride and then benzaldehyde from it.
Solution:

Toluene is first converted to benzal chloride by side-chain chlorination, in presence of Chlorine gas and light. Benzal chloride on hydrolysis at 373K gives benzaldehyde.
25. Name the electrophile produced in the reaction of benzene with benzoyl chloride in the presence of anhydrous AlCl3. Name the reaction also.
Solution:
The electrophile produced in the reaction of benzene with benzoyl chloride in the presence of anhydrous AlCl3 is benzoylinium cation. The product formed in this react ion is benzophenone. This reaction is called Friedel Craft’s acylation reaction.
26. Oxidation of ketones involves carbon-carbon bond cleavage. Name the products formed on oxidation of 2, 5-dimethylhexan-3-one.
Solution:
The products formed on oxidation of 2, 5-dimethylhexan-3-one are the mixtures of ketone and carboxylic acids. Ketone is then further oxidized to carboxylic acids. Overall the products formed in this reaction are 2-Methylpropanoic acid, 3-Methylbutanoic acid, ethanoic acid and methanoic acid.
27. Arrange the following in decreasing order of their acidic strength and give the reason for your answer.CH3CH2OH, CH3COOH, ClCH2COOH, FCH2COOH, C6H5CH2COOH
Solution:
FCH2COOH > ClCH2COOH > C6H5CH2COOH > CH3COOH > CH3CH2OH.
CH3CH2OH is least acidic among the given compounds. C6H5CH2COOH is more acidic than CH3COOH due to the resonance in C6H5CH2COOH. FCH2COOH and ClCH2COOH are highly acidic due to the –I effect of halogen.
28. What product will be formed on reaction of propanal with 2-methyl propanal in the presence of NaOH? What products will be formed? Write the name of the reaction also.
Solution:
When propanal reacts with 2-methyl propanal in the presence of NaOH, the mixture of aldehydes are formed. Both the reactants have an alpha-hydrogen and hence, can undergo cross aldol reaction in the presence of NaOH.

29. Compound ‘A’ was prepared by oxidation of compound ‘B’ with alkaline KMnO4. Compound ‘A’ on reduction with lithium aluminium hydride gets converted back to compound ‘B’. When compound ‘A’ is heated with compound B in the presence of H2SO4 it produces the fruity smell of compound C to which family the compounds ‘A’, ‘B’ and ‘C’ belong to?
Solution:
- Compound ‘A’ belongs to the carboxylic acid.
- Compound ‘B’ belongs to alcohol.
- Compound ‘C’ belongs to an ester group.
30. Arrange the following in decreasing order of their acidic strength. Explain the arrangement. C6H5COOH, FCH2COOH, NO2CH2COOH
Solution:
NO2CH2COOH > FCH2COOH > C6H5COOH.
NO2CH2COOH is most acidic among the given three compounds. Electron withdrawing groups like -NO2, increases the acidity of carboxylic acids by stabilizing the conjugate base through resonance effects
31. Alkenes and carbonyl compounds both contain a π bond but alkenes show electrophilic addition reactions whereas carbonyl compounds show nucleophilic addition reactions. Explain.


Solution:
Both the compounds carbon atom is attached to the electronegative atom oxygen. Thus the oxygen pulls more shared pair of electron towards them and a partial positive charge will be acquired by carbon and a partial negative charge by oxygen. So carbonyl atom is attacked by a nucleophile.
32. Carboxylic acids contain carbonyl group but do not show the nucleophilic addition reaction like aldehydes or ketones. Why?
Solution:
The oxygen atom in carbonyl compound pull more shared pair of electron towards itself and so, carbon acquires partial positive charge and oxygen acquires partial negative charge in carbonyl compounds. So, the carbon in the carbonyl atom is attacked by a nucleophile. Due to the resonance, carboxylic acids contain carbonyl group do not show the nucleophilic addition reaction like aldehydes or ketones
33. Identify the compounds A, B and C in the following reaction.
Solution:
a) Compound A = CH3-MgBr
b) Compound B = CH3-COOH
c) Compound C = CH3COOCH3
34. Why are carboxylic acids more acidic than alcohols or phenols although all of them have a hydrogen atom attached to an oxygen atom (—O—H)?
Solution:
Due to the resonance in carboxylic acids, the negative charge is at the more electronegative oxygen whereas, in alcohols or phenols, the negative charge is on a less electronegative atom. Thus, carboxylic acids can release proton easier than alcohols or phenols.
35. Complete the following reaction sequence.

Solution:

36. Ethylbenzene is generally prepared by acetylation of benzene followed by reduction and not by direct alkylation. Think of a possible reason.
Solution:
This is due to the formation of polysubstituted products.
To avoid the formation of polysubstituted products Friedel-craft’s alkylation reaction is not used for the preparation of ethylbenzene but one can use Friedel-craft’s acylation reaction.
37. Can Gatterman-Koch reaction be considered similar to Friedel Craft’s acylation? Discuss.
Solution:
Both reactions resemble each other. In Friedel Craft’s acylation reaction, an aryl group or benzene is treated with an acid chloride in the presence of anhydrous AlCl3 and corresponding aldehyde or ketone is formed. In Gatterman-Koch reaction, benzene is treated with CO and HCl in the presence of AlCl3 and CuCl to yield benzaldehyde.
IV. Matching Type
Note: Match the items of Column I and Column II in the following questions.
38. Match the common names given in Column I with the IUPAC names given in
Column II
| Column I Common names
(i) Cinnamaldehyde (ii) Acetophenone (iii) Valeraldehyde (iv) Acrolein (v) Mesityl oxide |
Column II (IUPAC names)
(a) Pentanal (b) Prop-2-enal (c) 4-Methylpent-3-en-2-one (d) 3-Phenylprop-2-enal (e) 1-Phenylethanone |
|---|
Solution:
(i) is d
(ii) is e
(iii) is a
(iv) is b
(v) is c
39. Match the acids given in Column I with their correct IUPAC names given in
Column II.
| Column I (Acids)
(i) Phthalic acid (ii) Oxalic acid (iii) Succinic acid (iv) Adipic acid (v) Glutaric acid |
Column II (IUPAC names)
(a) Hexane-1,6-dioic acid (b) Benzene-1,2-dicarboxylic acid (c) Pentane-1,5-dioic acid (d) Butane-1,4-dioic acid (e) Ethane-1,2-dioic acid |
|---|
Solution:
(i) is b
(ii) is e
(iii) is d
(iv) is a
(v) is c
40. Match the reactions given in Column I with the suitable reagents given in
Column II.
| Column I (Reactions)
(i) Benzophenone → Diphenylmethane (ii) Benzaldehyde → 1-Phenylethanol (iii) Cyclohexanone → Cyclohexanol (iv) Phenyl benzoate → Benzaldehyde |
Column II(Reagents)
(a) LiAlH4 (b) DIBAL—H (c) Zn(Hg)/Conc. HCl (d) CH3MgBr |
|---|
Solution:
(i) is c
(ii) is d
(iii) is a
(iv) is b
41. Match the example given in Column I with the name of the reaction in Column II

Solution:
(i) is e
(ii) is d
(iii) is a
(iv) is b
(v) is f
(vi) is c
V. Assertion and Reason Type
Note: In the following questions a statement of assertion followed by a statement of reason is given. Choose the correct answer out of the following choices.
(i) Assertion and reason both are correct and the reason is the correct explanation of assertion.
(ii) Assertion and reason both are wrong statements.
(iii) The assertion is a correct statement but the reason is the wrong statement.
(iv) The assertion is a wrong statement but the reason is the correct statement.
(v) Assertion and reason both are correct statements but the reason is not the correct explanation of assertion.
42. Assertion: Formaldehyde is a planar molecule. Reason: It contains sp2 hybridised carbon atom.Solution:
Option (i) is correct.
43. Assertion: Compounds containing —CHO group are easily oxidised to corresponding carboxylic acids. Reason: Carboxylic acids can be reduced to alcohols by treatment with LiAlH4
Solution:
Option (v) is correct
44. Assertion: The α-hydrogen atom in carbonyl compounds is less acidic.
Reason: The anion formed after the loss of the α-hydrogen atom is resonance stabilised.
Solution:
Option (iv) is correct.
45. Assertion: Aromatic aldehydes and formaldehyde undergo Cannizaro reaction.
Reason: Aromatic aldehydes are almost as reactive as formaldehyde.Solution:
Option (iii) is correct.
46. Assertion: Aldehydes and ketones, both react with Tollen’s reagent to form a silver mirror.
Reason: Both, aldehydes and ketones contain a carbonyl group.
Solution:
Option (iv) is correct
NCERT Exemplar For Class 12 Science
The NCERT exemplars are an effective study material for scoring higher marks in the examination paper. Students must practise these additional questions for their own benefits, as these are curated by the best subject-matter experts to boost both knowledge and confidence. Students can easily access the ncert exemplar for class 12 science by visiting our website SimplyAcad and solve all the questions listed to secure maximum marks.
Here are some other NCERT exemplar for class 12 chemistry:
| NCERT exemplar for class 12 chemistry Chapter 1 | NCERT exemplar for class 12 chemistry Chapter 7 |
|---|---|
| NCERT exemplar for class 12 chemistry Chapter 2 | NCERT exemplar for class 12 chemistry Chapter 8 |
| NCERT exemplar for class 12 chemistry Chapter 3 | NCERT exemplar for class 12 chemistry Chapter 9 |
| NCERT exemplar for class 12 chemistry Chapter 4 | NCERT exemplar for class 12 chemistry Chapter 10 |
| NCERT exemplar for class 12 chemistry Chapter 5 | NCERT exemplar for class 12 chemistry Chapter 11 |
latest video
news via inbox
Nulla turp dis cursus. Integer liberos euismod pretium faucibua